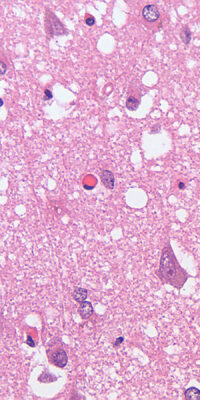
Prions are misfolded proteins that can cause fatal neurodegenerative diseases in animals and humans. They are highly resistant to inactivation and can persist in the environment for long periods of time. Prions pose a significant risk to animal research facilities, as they can contaminate equipment, surfaces, and biological materials, and transmit infections to experimental animals or humans.
Some of the most common prion diseases in animals are scrapie in sheep and goats, bovine spongiform encephalopathy (BSE) or mad cow disease in cattle, and chronic wasting disease (CWD) in deer and elk. These diseases are characterized by progressive neurological symptoms, such as behavioral changes, ataxia, tremors, and wasting, and are invariably fatal. Because there is no effective treatment or vaccine for prion diseases, deactivating prions within the environment is exceptionally important.
Transmission
The transmission of prions from animals to humans is rare but possible. The most well-known example is variant Creutzfeldt-Jakob disease (vCJD), which is caused by exposure to BSE prions from contaminated beef products. Other potential sources of human exposure include consumption of venison from CWD-infected cervids, contact with infected animal tissues or fluids, or use of medical products derived from animals.
Diagnosis
The diagnosis of prion diseases is challenging, as there are no reliable biomarkers or tests that can detect prions in living animals or humans. The definitive diagnosis can only be made by post-mortem examination of the brain tissue, which shows characteristic microscopic lesions of spongiform degeneration and accumulation of abnormal prion protein. However, some tests can provide presumptive evidence of prion infection, such as detection of abnormal prion protein in lymphoid tissues (e.g., tonsil or spleen) by immunohistochemistry or Western blot, or detection of proteinase-resistant prion protein in cerebrospinal fluid by real-time quaking-induced conversion (RT-QuIC).
Biosafety Measures
Prion research requires strict biosafety measures to prevent accidental exposure or transmission of prions. Human prions are manipulated at biosafety level (BSL) 2 or 3, depending on the activity, with most human prions treated as BSL-3 under most experimental conditions. BSE prions may also require the use of BSL-3 facilities due to the high probability that they have been transmitted to humans. All other animal prions are considered BSL-2 pathogens. However, when a prion from one species is inoculated into another, the resultant infected animal should be treated according to the guidelines applying to the source of the inoculum.
Need a prion disinfectant? In a recent study, Wex-cide 128, a phenolic disinfectant, was evaluated for efficacy against prions from various species compared to a similar product, Environ LpH (eLpH). The results indicate that Wex-cide 128 is highly effective against prions from hamsters, cervids, and mice, reducing infectivity by 4.0–5.5 logs. Because of this, Wex-Cide 128 emerges as a promising alternative for mitigating prion contamination across various species. Its strengths lie in providing a safer and more pragmatic choice for prion decontamination, particularly in research environments. You can learn more about Prion diseases, and Wex-Cide 128, by contacting your Biosafety Account Manager today.
—
References:
https://vetmedbiosci.colostate.edu/prc/
https://www.niaid.nih.gov/diseases-conditions/prion-research
https://ehs.msu.edu/lab-clinic/bio/handling-prions.html
https://policies.unc.edu/TDClient/2833/Portal/KB/ArticleDet?ID=131891
https://hsrm.umn.edu/biosafety-occupational-health/biosafety/working-safely-biological-materials-and-toxins/prions